Affordable Product for everyone.
Hand Gloves are a product of personal care for every one of us. We also have a variety of other medical things available.


Our Outstanding Product


GLOVE CARTON
PACKAGING CARTON DESIGN (1000PCS)


FACE MASK
3 PLY SURGICAL FACE MASK
SOON
MORE PRODUCTS


Our PRODUCT DESIGN
Gloves Singapore is in complies with the requirements.
The compliance with provisions of medical devices, including the following standards for requirement and testing for freedom from holes, physical properties, biological evaluation, terminology and performance requirements for chemical risks, determination of resistance to penetration, degradation by chemicals, for microorganisms risks, and permeation by liquid chemical vapors.
product specifications
Meeting with the Specification
Gloves are designed to accommodate both hands, and the inspection and testing procedures, as well as material inspection, are detailed.
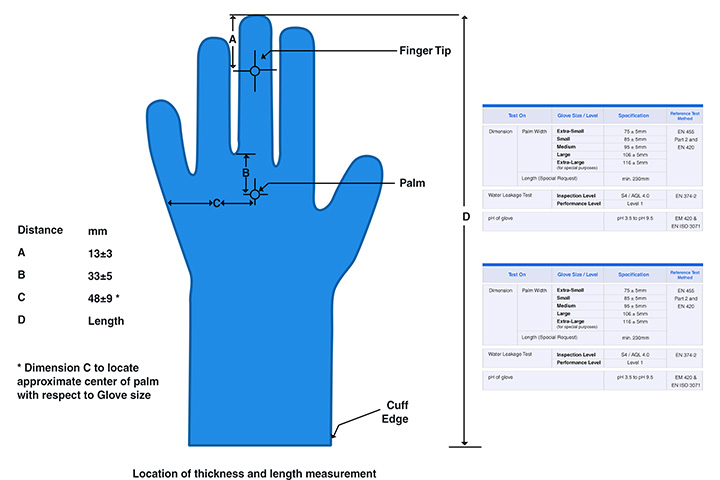

product specifications
Nitrile Examination Gloves
1.1. General Description
Glove Type
Glove: Powder-free Blue Nitrile Gloves (PFN-B)
Variants: Applicable for smooth or textured or finger-textured surface and meet the product specification.
Grade: AQL 4.0
Features: Ambidextrous, Disposable, Beaded Cuff
Material Made from: Natural Rubber
Colour: White
Shelf-Life: 3 years from date of manufacture
1.2. Intended Use
This disposable Personal Protective Equipment (PPE) rubber glove is intended to be work by an individual for protection against one or more health and safety hazards.
1.3. Classification
Category I: For minimal risk use only
1.4. Applicable Harmonised Standards
1.4.1. Meet EN 374 – part 1 and 2. Protective Gloves against water
1.5. Product Specification
The data sheet was made in collaboration with a consulting firm for use with the CE or FDA certificate.
Basic Health and Safety Requirements
2.1 General Requirements applicable to Powder-free Nitrile Glove PPE
1.1) The Gloves provide protection against:
- mechanical action whose effects are superficial (gardening glove).
- Cleaning materials of weak action and easily reversible effect (gloves affording protection against dilute detergent solution).
- Risk encountered in the handling of hot components which do not expose the user to a temperature exceeding 50°C or to dangerous impact
- Atmospheric agents of a neither exceptional nor extreme nature.
- Minor impact and vibration which do not affect the hands and whose effects cannot cause irreversible lesions.
1.2) Design of Gloves:
- Fit both hand, the gloved hands can perform the low risk-related activity whilst enjoying appropriate protection.
- The gloves materials and its decomposition products do not adversely affect user hygiene or health.
- The contact surface of the glove with the hand is free of roughness, sharp edges, projection and the likes which may cause excessive irritation or injuries.
- These gloves have good finger dexterity and do not impede the user movement or sensory perception.
1.3) Comfort and Efficiency
- 5 different sizes (Extra-Small, Small, Medium, Large and Extra Large) of the Gloves are supplied to ensure good fit on user’s hands morphology.
- The gloves are light, yet strong under foreseeable condition of use. It offers better puncture resistance than similar natural rubber latex glove.
- The pH of the gloves are close to neutral and do not cause irritation.
- These gloves are recommended for users that are allergic to natural rubber.
1.4) Information on the PPE device
- “Manufactured for” and address
- Storage condition of box
- Keep away from moisture.
- Keep away from heat (sun of fluorescence light).
- Store at 10-40°C
- Disposable
- The Gloves meet EN 374 – part 1 and 2
- There is no accessory associated with this PPE.
- Category I: Use for minimal risks condition (Laboratory, gardening, general cleaning at temperature of 10 – 50°C).
- The obsolescence deadline is 3 years from date of manufacture.
- For transportation, the gloves are packed in Paper Dispenser Boxes/Plastic Bag. A paper carton then encloses these Dispenser packages.
- The “Low Chemical Risk and Waterproof” pictograms are placed on the Dispenser Packages (refer section 7) to signify that the gloves were not tested against chemicals.
- The gloves comply with “Council Directive of 21 December 1989 relating to Personal Protective Equipment, 89/686/EEC.
- A notified body is not required to assess and verify the minimal risk PPE.
2.2. Additional Requirements for PPE Glove
2.2.1.) A bead is provided on the glove cuff to aid in donning of the hand.
2.2.2.) The date of obsolescence, 3 years after manufacturing is affixed on the dispenser boxes as the glove deteriorates slightly on aging. The useful life of the gloves can be prolonged with careful storage (refer clause 1.4 above).
2.2.3.) The glove can be torn by a sufficiently large force, thereby preventing it from being caught-up by a moving object.
2.2.4.) The gloves are ambidextrous in design, so it is easily don on the left or right hands. This also minimizes the time to don the gloves.
2.2.5.) The Pictograms “ Low Chemical Risk and Waterproof” are placed on the dispenser packages to indicate the risk that the user is protected against.
2.3. Additional Requirements Specific To Particular Risks.
2.3.1.) Protection against infective agents
- The glove when worn is capable of protecting the user hands against infective agent (micro-organism) as encountered in general use.
- The leak-tighness of the glove does not change on prolong usage, except
- Changed.
SECTION 4.0 QUALITY CONTROL SYSTEM
4.1 Incoming Material Inspection
4.1.1. All Incoming raw materials (that will affect the product’s quality) that are subjected to inspection are placed under “Quarantine” area in the store.
4.1.2. Incoming materials are selected randomly as per ISO 2859 sampling plan and inspected by INcoming Material Inspector.
4.1.3. Incoming materials that meet In-house specifications are attached with “Passed” sticker.
4.1.4. Incoming materials that do not meet in-house specifications are quarantined pending management disposition decision.
- The Incoming materials are needed to be screened are attached with “Rejected” sticker and with “SCREEN” stamp on it.
- The Incoming materials that can be “Used as it” is (substandard material but without functional implication and actual affect on quality) are attached with “Rejected” sticker and with “UAI” stamp on it.
- Incoming materials that cannot used and needed to be returned back to supplier are attached with “Reject” sticker.
4.1.5. All Reject Materials are return immediately to Supplier or stored in “Reject Area” pending return to supplier.
4.2 Milling and Compounding of Raw Material
4.2.1. Only the materials that with “Passed” sticker can be used for milling process.
4.2.2. The milled chemical is analyzed by laboratory personnel.
4.2.3. Only the milled chemical that has passed laboratory analysis will be released to be used in Compounding process.
4.2.4. The milled chemical that has failed laboratory analysis shall be re-milled and quarantined to be used until it passes laboratory analysis.
4.2.5. Only the approved milled chemicals and chemicals with “Passes” sticker are used for compounding process.
4.2.6. The nitrile compounds are analyzed by laboratory personnel.
4.2.7. The nitrile compounds that pass all the in-house specification will be released to be use in production.
4.2.8. The nitrile compounds that fail the In-house specification will be quarantined to be used and shall wait for the management disposition decision.
4.2.9. All adjustment made on the nitrile compounds will be documented in a work sheet and shall be analyzed by laboratory personnel again.
4.3. Production On-line Goods
4.3.1.Each bin of gloves is issued with a Travel Card and is controlled to 8 kgs.
4..3.2. Samples are taken and inspected as per ISO 2859 table I and II-A
4.3.3. Every 6 bins, grouped as one Lot shall be tested as follow;
Inspect On Inspection Level AQL
i) Pin-hole 80 pcs 0
ii) Dimension S2 5
iii) Visual Major 80 pcs 4.0
iv) Visual Minor 80 pcs 5
4.3.4. The bins that have passed the above inspection will be marked as grade AQL 4.0. On the Travel card.
4.3.5. The bins that had failed the above inspection will be segregated and quarantined and stored in a specified area in store.
4.3.6. The passed bins will proceed to WIP Store or Packing Department.
4.4. Packing QC Inspection
4.4.1. At packing, Packing QC conduct line clearance inspection before commence packing and at every hourly.
4.5 Finished Goods
4.5.1. The pallets of finish packed gloves are inspected by pre-shipment QA.
4.5.2. The samples are randomly picked and inspected as per ISO 2859 table I and II-A for;
Inspect On Inspection Level AQL
- i) Pin-hole (watertight) S4 0
- ii) Dimension S2 0
iii) Visual Major S4 4.0
- iv) Visual Minor S4 5
- v) pH of Glove N=3 –
4.5.3. The pallets of packed gloves that have passed Pre-shipment QA Inspection are ready to deliver and store in outgoing store.
4.5.4. The pallets of packed gloves that have failed Pre-shipment QA Inspection shall be reworked by Packing Department.
4.5.5. The sorted gloves will be quarantined and stored in specified area and wait for management disposition decision.
5.1 Description of the Control and Test Facilities.
Area Control and Test Facilities
- Laboratory
- Chemical concentration testing apparatus
- Quality Control (QC) Section
- Water-tight Test Equipment.
- Stainless Steel Ruler.
- Thickness Gauge.
- Pre-Packing QA Section
- Water-tight Test Equipment
- Stainless Steel Ruler.
- Pre-shipment
- 1) Water-tight Test Equipment.
- 2) Stainless Steel Ruler.
- 3) Thickness Gauge.
SECTION 6.0 PRODUCT TESTING
Test were conducted to assure that the safe and correct operation of the product maintained as well as achieving the performance and compatibilities as required by the Directive. The Test results include as attached:
- Skin Irritation Test
- Dermal Sensitization Test
- Chemical Residue Test
- Product Test Report
SECTION 7.0 LIST OF PICTOGRAMS (For Labelling)
7.1 The Pictograms listed below can be replaced by suitable wordings, if necessary;
7.2. Refer attachment for a sample of dispenser labeling.
SECTION 8.0 LIST OF STANDARDS APPLIED
8.1. The referenced harmonized standards applied
- EN 374-1:2003, Protective gloves against chemicals and micro-organism –
Part 1: Terminology and performance requirements. - EN 374-2:2003, Protective gloves against chemicals and micro-organism –
Part 2: Determination of resistance to penetration. - EN 420: 2003 + A1: 2009, Protective gloves – General requirements and test methods.
- EN 455-2: 2009 Medical gloves for single use – Part 2: Requirements and testing for physical properties.
- EN 455-3: 2006, Medical gloves for single use – Part 3: Requirements and testing for biological evaluation.
- EN ISO 3071: 2006 Textiles – Determination of pH of aqueous extract.
- ISO 2859-1:1999, Sampling procedures for inspection by attributes –
Part 1: Sampling schemes indexed by acceptance quality limit (AQL) for lot-by-lot inspection. - ISO 9001:2008, Quality management systems – Requirements. (Certificate attached)
- ISO 10993-1:2003, Biological evaluation of medical devices –
Part: 1 Evaluation and Testing. - ISO 10993-10: 2002, Biological evaluation of medical devices –
Part: 10 Test for irritation and sensitization.
product specifications
Latex Examination Gloves
1.1. General Description
Glove Type
Glove: Powder-free Blue Nitrile Gloves (PFN-B)
Variants: Applicable for smooth or textured or finger-textured surface and meet the product specification.
Grade: AQL 4.0
Features: Ambidextrous, Disposable, Beaded Cuff
Material Made from: Synthetic Nitrile Rubber
Colour: Blue colored
Shelf-Life: 3 years from date of manufacture
1.2. Intended Use
This disposable Personal Protective Equipment (PPE) rubber glove is intended to be work by an individual for protection against one or more health and safety hazards.
1.3. Classification
Category I: For minimal risk use only
1.4. Applicable Harmonised Standards
1.4.1. Meet EN 374 – part 1 and 2. Protective Gloves against water
1.5. Product Specification
The data sheet was made in collaboration with a consulting firm for use with the CE or FDA certificate.
Basic Health and Safety Requirements
2.1 General Requirements applicable to Powder-free Nitrile Glove PPE
1.1) The Gloves provide protection against:
- mechanical action whose effects are superficial (gardening glove).
- Cleaning materials of weak action and easily reversible effect (gloves affording protection against dilute detergent solution).
- Risk encountered in the handling of hot components which do not expose the user to a temperature exceeding 50°C or to dangerous impact
- Atmospheric agents of a neither exceptional nor extreme nature.
- Minor impact and vibration which do not affect the hands and whose effects cannot cause irreversible lesions.
1.2) Design of Gloves:
- Fit both hand, the gloved hands can perform the low risk-related activity whilst enjoying appropriate protection.
- The gloves materials and its decomposition products do not adversely affect user hygiene or health.
- The contact surface of the glove with the hand is free of roughness, sharp edges, projection and the likes which may cause excessive irritation or injuries.
- These gloves have good finger dexterity and do not impede the user movement or sensory perception.
1.3) Comfort and Efficiency
- 5 different sizes (Extra-Small, Small, Medium, Large and Extra Large) of the Gloves are supplied to ensure good fit on user’s hands morphology.
- The gloves are light, yet strong under foreseeable condition of use. It offers better puncture resistance than similar natural rubber latex glove.
- The pH of the gloves are close to neutral and do not cause irritation.
- These gloves are recommended for users that are allergic to natural rubber.
1.4) Information on the PPE device
- “Manufactured for” and address
- Storage condition of box
- Keep away from moisture.
- Keep away from heat (sun of fluorescence light).
- Store at 10-40°C
- Disposable
- The Gloves meet EN 374 – part 1 and 2
- There is no accessory associated with this PPE.
- Category I: Use for minimal risks condition (Laboratory, gardening, general cleaning at temperature of 10 – 50°C).
- The obsolescence deadline is 3 years from date of manufacture.
- For transportation, the gloves are packed in Paper Dispenser Boxes/Plastic Bag. A paper carton then encloses these Dispenser packages.
- The “Low Chemical Risk and Waterproof” pictograms are placed on the Dispenser Packages (refer section 7) to signify that the gloves were not tested against chemicals.
- The gloves comply with “Council Directive of 21 December 1989 relating to Personal Protective Equipment, 89/686/EEC.
- A notified body is not required to assess and verify the minimal risk PPE.
2.2. Additional Requirements for PPE Glove
2.2.1.) A bead is provided on the glove cuff to aid in donning of the hand.
2.2.2.) The date of obsolescence, 3 years after manufacturing is affixed on the dispenser boxes as the glove deteriorates slightly on aging. The useful life of the gloves can be prolonged with careful storage (refer clause 1.4 above).
2.2.3.) The glove can be torn by a sufficiently large force, thereby preventing it from being caught-up by a moving object.
2.2.4.) The gloves are ambidextrous in design, so it is easily don on the left or right hands. This also minimizes the time to don the gloves.
2.2.5.) The Pictograms “ Low Chemical Risk and Waterproof” are placed on the dispenser packages to indicate the risk that the user is protected against.
2.3. Additional Requirements Specific To Particular Risks.
2.3.1.) Protection against infective agents
- The glove when worn is capable of protecting the user hands against infective agent (micro-organism) as encountered in general use.
- The leak-tighness of the glove does not change on prolong usage, except
- Changed.
SECTION 4.0 QUALITY CONTROL SYSTEM
4.1 Incoming Material Inspection
4.1.1. All Incoming raw materials (that will affect the product’s quality) that are subjected to inspection are placed under “Quarantine” area in the store.
4.1.2. Incoming materials are selected randomly as per ISO 2859 sampling plan and inspected by INcoming Material Inspector.
4.1.3. Incoming materials that meet In-house specifications are attached with “Passed” sticker.
4.1.4. Incoming materials that do not meet in-house specifications are quarantined pending management disposition decision.
- The Incoming materials are needed to be screened are attached with “Rejected” sticker and with “SCREEN” stamp on it.
- The Incoming materials that can be “Used as it” is (substandard material but without functional implication and actual affect on quality) are attached with “Rejected” sticker and with “UAI” stamp on it.
- Incoming materials that cannot used and needed to be returned back to supplier are attached with “Reject” sticker.
4.1.5. All Reject Materials are return immediately to Supplier or stored in “Reject Area” pending return to supplier.
4.2 Milling and Compounding of Raw Material
4.2.1. Only the materials that with “Passed” sticker can be used for milling process.
4.2.2. The milled chemical is analyzed by laboratory personnel.
4.2.3. Only the milled chemical that has passed laboratory analysis will be released to be used in Compounding process.
4.2.4. The milled chemical that has failed laboratory analysis shall be re-milled and quarantined to be used until it passes laboratory analysis.
4.2.5. Only the approved milled chemicals and chemicals with “Passes” sticker are used for compounding process.
4.2.6. The nitrile compounds are analyzed by laboratory personnel.
4.2.7. The nitrile compounds that pass all the in-house specification will be released to be use in production.
4.2.8. The nitrile compounds that fail the In-house specification will be quarantined to be used and shall wait for the management disposition decision.
4.2.9. All adjustment made on the nitrile compounds will be documented in a work sheet and shall be analyzed by laboratory personnel again.
4.3. Production On-line Goods
4.3.1.Each bin of gloves is issued with a Travel Card and is controlled to 8 kgs.
4..3.2. Samples are taken and inspected as per ISO 2859 table I and II-A
4.3.3. Every 6 bins, grouped as one Lot shall be tested as follow;
Inspect On Inspection Level AQL
i) Pin-hole 80 pcs 0
ii) Dimension S2 5
iii) Visual Major 80 pcs 4.0
iv) Visual Minor 80 pcs 5
4.3.4. The bins that have passed the above inspection will be marked as grade AQL 4.0. On the Travel card.
4.3.5. The bins that had failed the above inspection will be segregated and quarantined and stored in a specified area in store.
4.3.6. The passed bins will proceed to WIP Store or Packing Department.
4.4. Packing QC Inspection
4.4.1. At packing, Packing QC conduct line clearance inspection before commence packing and at every hourly.
4.5 Finished Goods
4.5.1. The pallets of finish packed gloves are inspected by pre-shipment QA.
4.5.2. The samples are randomly picked and inspected as per ISO 2859 table I and II-A for;
Inspect On Inspection Level AQL
- i) Pin-hole (watertight) S4 0
- ii) Dimension S2 0
iii) Visual Major S4 4.0
- iv) Visual Minor S4 5
- v) pH of Glove N=3 –
4.5.3. The pallets of packed gloves that have passed Pre-shipment QA Inspection are ready to deliver and store in outgoing store.
4.5.4. The pallets of packed gloves that have failed Pre-shipment QA Inspection shall be reworked by Packing Department.
4.5.5. The sorted gloves will be quarantined and stored in specified area and wait for management disposition decision.
5.1 Description of the Control and Test Facilities.
Area Control and Test Facilities
- Laboratory
- Chemical concentration testing apparatus
- Quality Control (QC) Section
- Water-tight Test Equipment.
- Stainless Steel Ruler.
- Thickness Gauge.
- Pre-Packing QA Section
- Water-tight Test Equipment
- Stainless Steel Ruler.
- Pre-shipment
- 1) Water-tight Test Equipment.
- 2) Stainless Steel Ruler.
- 3) Thickness Gauge.
SECTION 6.0 PRODUCT TESTING
Test were conducted to assure that the safe and correct operation of the product maintained as well as achieving the performance and compatibilities as required by the Directive. The Test results include as attached:
- Skin Irritation Test
- Dermal Sensitization Test
- Chemical Residue Test
- Product Test Report
SECTION 7.0 LIST OF PICTOGRAMS (For Labelling)
7.1 The Pictograms listed below can be replaced by suitable wordings, if necessary;
7.2. Refer attachment for a sample of dispenser labeling.
SECTION 8.0 LIST OF STANDARDS APPLIED
8.1. The referenced harmonized standards applied
- EN 374-1:2003, Protective gloves against chemicals and micro-organism –
Part 1: Terminology and performance requirements. - EN 374-2:2003, Protective gloves against chemicals and micro-organism –
Part 2: Determination of resistance to penetration. - EN 420: 2003 + A1: 2009, Protective gloves – General requirements and test methods.
- EN 455-2: 2009 Medical gloves for single use – Part 2: Requirements and testing for physical properties.
- EN 455-3: 2006, Medical gloves for single use – Part 3: Requirements and testing for biological evaluation.
- EN ISO 3071: 2006 Textiles – Determination of pH of aqueous extract.
- ISO 2859-1:1999, Sampling procedures for inspection by attributes –
Part 1: Sampling schemes indexed by acceptance quality limit (AQL) for lot-by-lot inspection. - ISO 9001:2008, Quality management systems – Requirements. (Certificate attached)
- ISO 10993-1:2003, Biological evaluation of medical devices –
Part: 1 Evaluation and Testing. - ISO 10993-10: 2002, Biological evaluation of medical devices –
Part: 10 Test for irritation and sensitization.